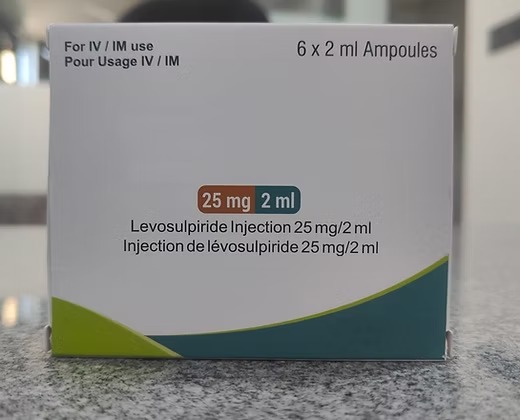
What is Levosulpiride Injection?
Levosulpiride is the levo-enantiomer of sulpiride, which enhances gastric emptying and possesses antipsychotic and antidepressant properties. In injectable form, it is commonly used in hospitals for rapid symptomatic relief of nausea, vomiting, gastritis, GERD, and certain psychiatric conditions.
Key Uses of Levosulpiride Injection
- Gastroparesis (delayed stomach emptying)
- Functional dyspepsia
- Gastroesophageal reflux disease (GERD)
- Irritable bowel syndrome (IBS)
- Chronic gastritis
How Does Levosulpiride Injection Works
Levosulpiride acts by selectively blocking dopamine D2 receptors both in the central nervous system and enteric nervous system. This leads to:
- Enhanced gastrointestinal motility
- Reduced nausea and vomiting
- Modulation of mood and anxiety symptoms (in higher doses)
Common Side Effects of Levosulpiride Injection
- Drowsiness or sedation
- Weight gain
- Galactorrhea or menstrual irregularities (due to hyperprolactinemia)
- Dizziness or extrapyramidal symptoms (in higher doses)
- Injection site discomfort
Use with caution in Parkinson’s disease, epilepsy, or patients with prolactin-dependent tumors.
Why Choose Farbe Firma for Levosulpiride Injection?
WHO-GMP Certified Injectable Manufacturing
Produced under stringent quality controls using sterile, modern facilities.
Comprehensive Regulatory Support
CTD dossier available, including FSC, COA, COPP, MSDS, and stability data.
Export Presence in 30+ Countries
Active markets in Africa, Asia, CIS, and Latin America.
Custom Manufacturing & Private Labeling
Flexible MOQs, contract manufacturing, and buyer branding support.
Timely Delivery & Competitive Pricing
Robust supply chain with strong export packaging & documentation support
FAQs on Levosulpiride Injection
Q1: What is Levosulpiride Injection used for?
Used to treat acid reflux, gastric motility disorders, nausea, and as an adjunct in some psychotic disorders.
Q2: How is it administered?
Administered via intramuscular (IM) or intravenous (IV) injection, under medical supervision.
Q3: What is the shelf life?
Typically 24 to 36 months, depending on packaging and storage conditions.
Q4: Do you offer documentation and samples for tenders or registration?
Yes. Farbe Firma provides samples, CTD-format dossiers, and regulatory support for global tenders and filings.
Looking for a Reliable Levosulpiride Injection Manufacturer & Exporter?
Partner with Farbe Firma – a globally trusted name for gastrointestinal, neurological, and hospital-use injectables.