What is Cerebroprotein Hydrolysate Injection?
Cerebroprotein Hydrolysate Injection is a sterile injectable solution made from purified porcine brain-derived peptides. It mimics the action of naturally occurring neurotrophic factors that stimulate nerve regeneration, neuroplasticity, and protect neurons from damage.
It is widely used in neurology and rehabilitation medicine for managing acute and chronic brain-related disorders.
Uses of Cerebroprotein Hydrolysate Injection
Cerebroprotein Hydrolysate is indicated for the treatment of:
- Ischemic stroke (acute and recovery phase)
- Traumatic brain injury (TBI)
- Alzheimer’s disease and other dementias
- Parkinson’s disease (adjunct therapy)
- Cognitive decline due to aging
- Memory loss and attention disorders
- Cerebral palsy (in pediatric use, under specialist supervision)
- Post-neurosurgical rehabilitation
This injection supports brain recovery and improves neurocognitive performance in patients with impaired brain functions.
How Does Cerebroprotein Hydrolysate Injection Work?
Cerebroprotein Hydrolysate contains low molecular weight peptides that cross the blood-brain barrier and:
- Stimulate neuronal growth and repair
- Improve synaptic plasticity and memory
- Reduce oxidative stress and inflammation
- Protect neurons from excitotoxicity and apoptosis
- Support neurotransmitter function
These actions collectively enhance brain recovery, function, and neuroprotection, especially in post-stroke and degenerative conditions.
Common Side Effects of Cerebroprotein Hydrolysate Injection
While it is generally well-tolerated, some patients may experience:
- Headache
- Nausea
- Mild fever or chills
- Local injection site reactions
- Dizziness
- Restlessness or insomnia (rare)
Contraindications: Not recommended during pregnancy, lactation, or in patients with epilepsy unless supervised by a physician.
Why Choose farbe firma for Cerebroprotein Hydrolysate Injection?
farbe firma is a trusted manufacturer and exporter of high-quality injectable formulations, offering custom packaging, low MOQs, and regulatory support.
WHO GMP Certified Manufacturing
All products are manufactured in compliance with WHO-GMP, ISO, and local regulatory norms.
Modern Injectable Infrastructure
State-of-the-art facilities for ampoules and vials, ensuring sterile, stable, and safe neuroinjectables.
Private Label & Contract Manufacturing
We offer full contract manufacturing, custom branding, and private label services tailored to your market.
Global Export Capabilities
We supply Cerebroprotein Hydrolysate Injection to 30+ countries across Africa, CIS, Asia-Pacific, Latin America, and more, with CTD/ACTD dossier support.
FAQs on Cerebroprotein Hydrolysate Injection
Q1: What is Cerebroprotein Hydrolysate Injection used for?
It is used to treat neurological disorders such as stroke, brain injury, Alzheimer’s, and age-related cognitive decline by improving brain cell function and regeneration.
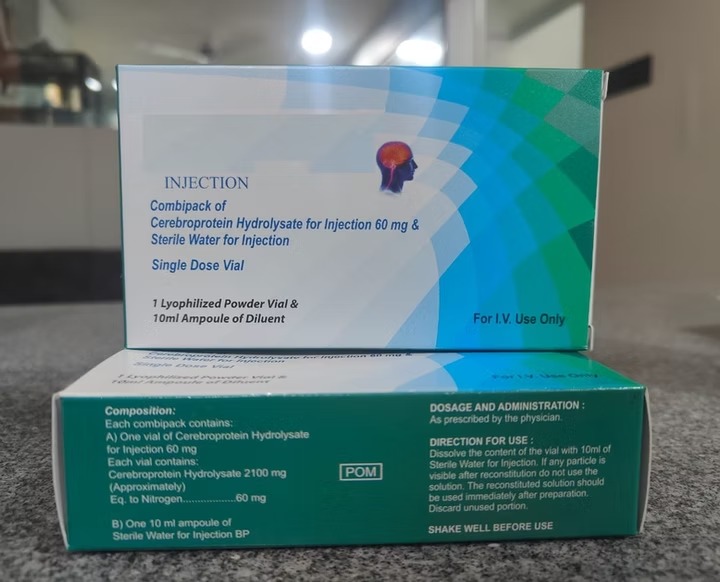
Q2: What are the available strengths and packaging?
Typically available in 60 mg/3 ml and 90 mg/3 ml ampoules or vials. Custom formulations are available for bulk orders.
Q3: Do you offer regulatory and export documentation?
Yes, we provide complete CTD/ACTD dossiers, COA, MSDS, and stability data for registration in various countries.
Q4: Can you provide product samples for evaluation?
Yes. Samples are available upon request for qualified partners, subject to regulatory compliance.
Q5: What is the shelf life of this product?
Standard shelf life is 24 to 36 months, depending on formulation and packaging.
Looking for a Trusted Manufacturer & Exporter of Cerebroprotein Hydrolysate Injection?
Partner with farbe firma for reliable supply, regulatory support, and high-quality neuroinjectables.
- WHO-GMP certified injectable manufacturing
- Flexible MOQ & custom branding
- Full export documentation & shipping
- Global delivery to 30+ countries
📞 Get in touch today to request a quote or sample.