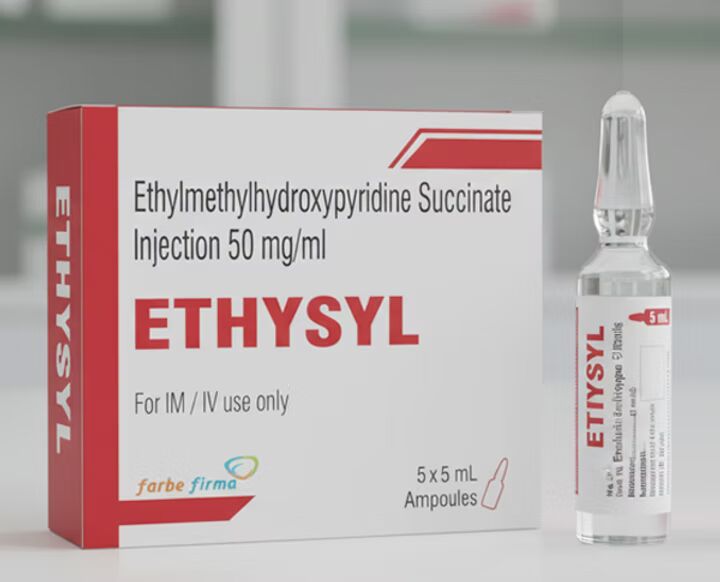
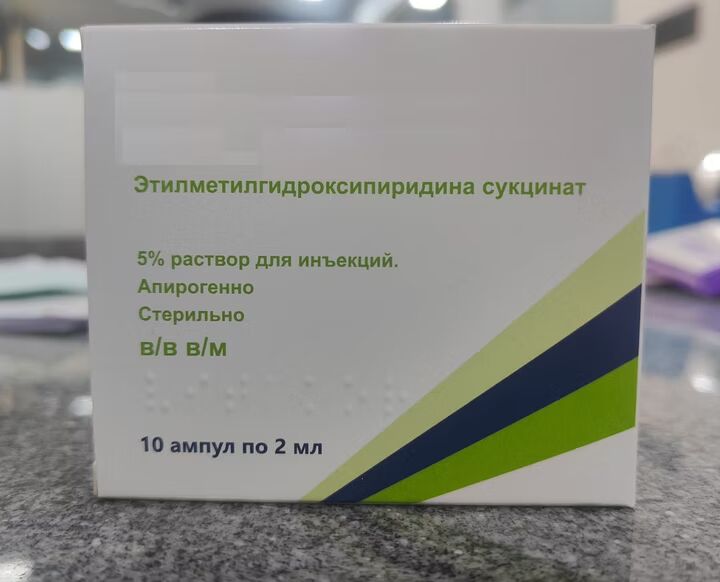
What is Ethylmethylhydroxypyridine Succinate Injection?
Ethylmethylhydroxypyridine Succinate Injection is a powerful antioxidant and neuroprotective agent used in the treatment of acute neurological and cardiovascular conditions. Salvavidas Pharma, a leading WHO GMP certified manufacturer, offers high-quality formulations of this injection for global markets with complete regulatory support and export services.
Eedthylmethylhydroxypyridine Succinate Injection is a sterile injectable solution primarily us in the management of neurological disorders, stroke, ischemic brain conditions, and oxidative stress-related complications.
Also known by the brand name Mexidol® in some countries, it exhibits antioxidant, antihypoxic, and membrane-stabilizing properties, helping protect nerve cells from damage during oxygen deprivation or stress.
Uses of Ethylmethylhydroxypyridine Succinate Injection
This injection is indicated for the treatment and prevention of:
- Acute ischemic stroke
- Traumatic brain injury (TBI)
- Cerebral edema
- Encephalopathy
- Neurodegenerative conditions
- Cognitive disorders
- Cardiovascular diseases
- Anxiety and oxidative stress
- Alcohol withdrawal syndrome
- Mild cognitive impairment due to aging
Often used in neurology, cardiology, and intensive care units.
How Does Ethylmethylhydroxypyridine Succinate Injection Work?
Ethylmethylhydroxypyridine Succinate is an antioxidant that neutralizes free radicals, reducing oxidative damage in brain and heart tissues. It:
- Improves oxygen delivery and cellular respiration
- Stabilizes cell membranes
- Reduces inflammation and neurotoxicity
- Enhances cognitive and neurological recovery
- Improves microcirculation and metabolic processes
Its neuroprotective effects make it valuable in emergency neurological interventions and long-term cognitive therapies.
Common Side Effects of Ethylmethylhydroxypyridine Succinate Injection
While generally well-tolerated, some patients may experience:
- Nausea or vomiting
- Dizziness or headache
- Allergic reactions (rash, itching)
- Sleep disturbances (rare)
- Mild drop in blood pressure
- Local irritation at injection site
Always administer under medical supervision. Not recommended during pregnancy, lactation, or in patients with severe liver or kidney dysfunction unless advised by a physician.
Why Choose Farbe Firma as Your Ethylmethylhydroxypyridine Succinate Injection?
farbe firma is a reputed Indian manufacturer and global exporter of high-quality injectable formulations, offering full support from production to international market distribution.
WHO-GMP Compliant Manufacturing Facility
Ensuring international-quality injectable production in sterile, controlled environments.
Flexible Packaging & Dosage Options
Available in ampoules or vials with customizable strengths to meet market-specific needs.
Complete Contract Manufacturing Support
We offer private labeling, custom branding, and full contract manufacturing services for global partners.
Global Export to 30+ Countries
Strong export presence in Africa, CIS, Southeast Asia, Latin America, and Middle East with CTD/ACTD dossier support and product registration assistance.
FAQs on Ethylmethylhydroxypyridine Succinate Injection
Q1: What is Ethylmethylhydroxypyridine Succinate Injection used for?
It is used in the treatment of stroke, brain injuries, neurological disorders, and cardiovascular diseases due to its neuroprotective and antioxidant properties.
Q2: What is the standard dosage form and strength?
Typically available as 100 mg/2 ml in ampoule form, or customized as per buyer requirements.
Q3: Is this product suitable for export markets?
Yes. Salvavidas Pharma supplies this product to international markets with dossier support, stability data, and GMP certifications.
Q4: What is the shelf life of the product?
Usually 24–36 months under standard storage conditions. Specific shelf life depends on packaging and market regulations.
Q5: Can I request product samples and documents?
Yes. We provide samples, COAs, MOAs, and registration documents upon request, subject to terms.
Looking for a Reliable Ethylmethylhydroxypyridine Succinate Injection
Choose Salvavidas Pharma for quality, consistency, and compliance.
- WHO-GMP certified production
- Export to 30+ countries
- Low MOQ & bulk supply
- Complete regulatory and logistic support
📞 Get in touch today to request a quote or sample.