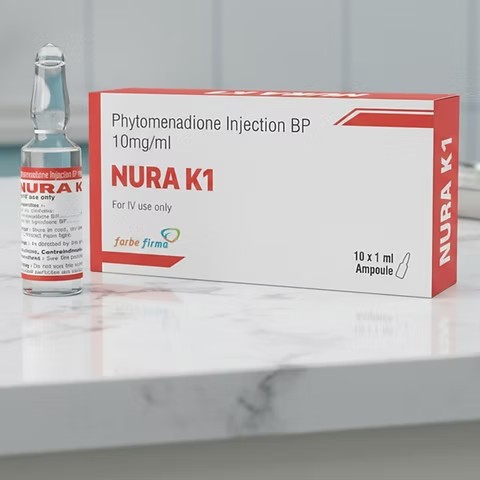
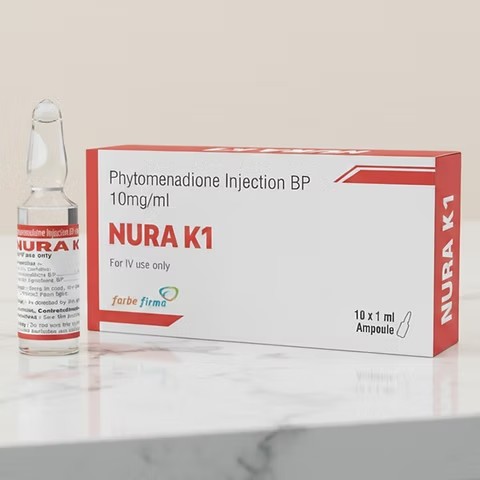
What is Phytomenadione Injection (Vitamin K1)?
Phytomenadione Injection is a sterile injectable solution of Vitamin K1, a fat-soluble vitamin critical for synthesizing blood-clotting factors. It is used to correct or prevent bleeding disorders due to low levels of vitamin K in the body.
Uses of Phytomenadione Injection
- Prevention and treatment of vitamin K deficiency bleeding (VKDB) in newborns
- Reversal of anticoagulant (e.g., warfarin) overdose
- Management of hypoprothrombinemia caused by liver disease, malabsorption, or antibiotic therapy
- Treatment of bleeding disorders caused by prolonged use of certain medications
How Does Phytomenadione Injection Work?
Phytomenadione (Vitamin K1) aids in the activation of clotting factors II, VII, IX, and X in the liver. It restores normal clotting function in patients with vitamin K deficiency, effectively stopping or preventing bleeding episodes.
Common Side Effects of Phytomenadione Injection
- Pain, redness, or swelling at the injection site
- Flushing or dizziness
- Changes in taste
- Rare but serious allergic reactions (e.g., anaphylaxis)
- Hypersensitivity reactions (especially with IV administration)
Why Choose farbe firma for Phytomenadione Injection (Vitamin K1)?
WHO-GMP Certified Manufacturing Facility
Manufactured in ISO & WHO-GMP approved sterile injectable plants with strict quality controls.
Global Export Expertise
Supplying to 30+ countries including Africa, Latin America, CIS, and Southeast Asia.
Full Regulatory & Documentation Support
Ideal for tenders, distributors, and hospitals with low minimum order quantities and custom branding support.
End-to-End Contract Manufacturing Services
From formulation development to finished product delivery, tailored to your market needs.
FAQs on Phytomenadione Injection (Vitamin K1)
Q1: What is Phytomenadione Injection used for?
It is used to prevent or treat bleeding caused by vitamin K deficiency in newborns, adults, and patients on anticoagulant therapy.
Q2: How is it administered?
It can be given intravenously (IV), intramuscularly (IM), or subcutaneously, depending on the clinical need.
Q3: What is the shelf life?
Typically 24 to 36 months when stored in cool, dark conditions as recommended.
Q4: Do you provide dossiers and export documentation?
Yes, complete regulatory documentation is available for product registration in multiple countries.
Looking for a Reliable Phytomenadione Injection Manufacturer & Exporter?
Choose Farbe Firma for:
- WHO-GMP certified injectable manufacturing
- Full regulatory support for international tenders
- Fast lead times & flexible packaging solutions
- Competitive pricing for bulk or contract manufacturing
Contact us Get in touch today for pricing, samples, or registration dossiers.