What is Tramadol Hydrochloride Injection?
Tramadol Hydrochloride Injection is a sterile, parenteral opioid analgesic formulation used to relieve acute and chronic pain. It acts on the central nervous system (CNS) to change the body’s response to pain signals.
It is commonly used in post-operative care, injury-related pain, orthopedic conditions, and other painful procedures when oral administration is not feasible..
Uses of Tramadol Hydrochloride Injection
Tramadol Injection is indicated for:
- Moderate to severe pain management
- Post-operative pain
- Pain due to trauma or fractures
- Cancer-related pain
- Chronic musculoskeletal pain
- Pain during diagnostic procedures
It is administered intravenously (IV) or intramuscularly (IM) in clinical and emergency settings under medical supervision.
How Does Tramadol Hydrochloride Injection Work?
Tramadol works by binding to mu-opioid receptors in the brain and inhibiting the reuptake of norepinephrine and serotonin, which modulates the perception of pain.
Its dual mechanism of action provides effective pain relief with a lower risk of respiratory depression compared to other opioids.
Common Side Effects of Tramadol Hydrochloride Injection
Tramadol is generally well-tolerated, but potential side effects include:
- Nausea and vomiting
- Dizziness or lightheadedness
- Sweating
- Constipation
- Drowsiness or sedation
- Headache
- Itching or rash (less common)
Serious but rare side effects:
- Respiratory depression (especially at high doses)
- Seizures
- Serotonin syndrome (if used with serotonergic drugs)
- Dependence with long-term use
Note: Should be used with caution in patients with renal or hepatic impairment, history of seizures, or substance abuse.
Why Choose farbe firma for Tramadol Hydrochloride Injection?
farbe firma is a trusted name in injectable formulations, offering high-quality, regulatory-compliant Tramadol Injections for institutional use and export markets.
WHO GMP Certified Manufacturing
All injectables are produced under stringent WHO-GMP and ISO guidelines.
Flexible Strengths and Packaging
Common presentations include:
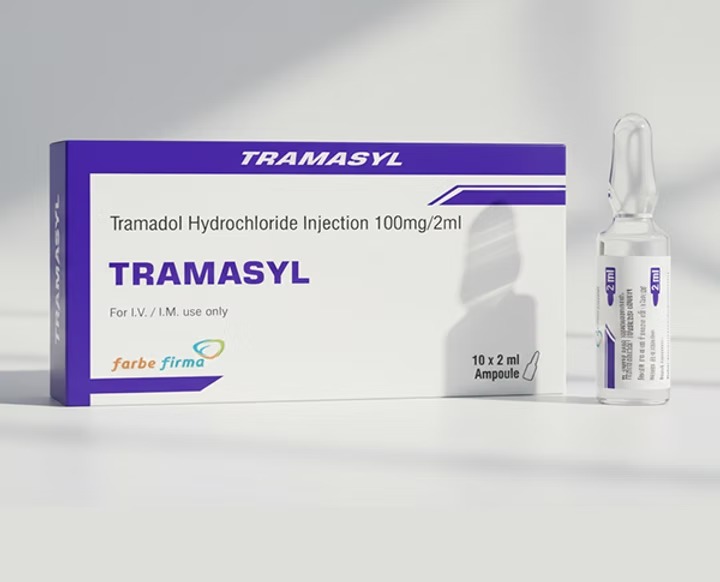
- Tramadol 50 mg/ml – 1 ml ampoule
- Tramadol 100 mg/2 ml – ampoule or vial
Custom packaging (ampoules, vials, boxes) available for private label/contract manufacturing.
Global Export Expertise
Exporting to 30+ countries including Africa, CIS, Asia-Pacific, and Latin America, with complete dossier and product registration support.
Full Contract Manufacturing & Private Label Support
Low MOQ, flexible branding, and export-ready formulations with stability data and documentation.
FAQs on Tramadol Hydrochloride Injection
Q1: What is Tramadol Injection used for?
It is used to treat moderate to severe pain in post-operative, trauma, or chronic conditions when oral painkillers are not suitable.
Q2: What strengths are available?
Standard strengths include:
- 50 mg/ml (1 ml ampoule)
- 100 mg/2 ml ampoule
Custom strengths/formulations available upon request.
Q3: What is the shelf life?
Typical shelf life is 24–36 months, depending on packaging and storage conditions.
Q4: Is this product available for export?
Yes. We offer Tramadol Hydrochloride Injection with CTD/ACTD dossiers, COAs, and stability data for international registration.
Q5: Can I get samples for evaluation?
Yes, samples are available upon request, subject to regulatory and commercial terms.
Looking for a Reliable Manufacturer & Exporter of Tramadol Hydrochloride Injection?
Partner with farbe firma for a dependable supply of high-quality Tramadol Injections supported by:
- WHO-GMP manufacturing
- Low MOQ & bulk supply
- Regulatory documentation
- Fast, compliant export logistics
Contact us now for quotes, samples, or regulatory support.